组内消息
2024-11-15
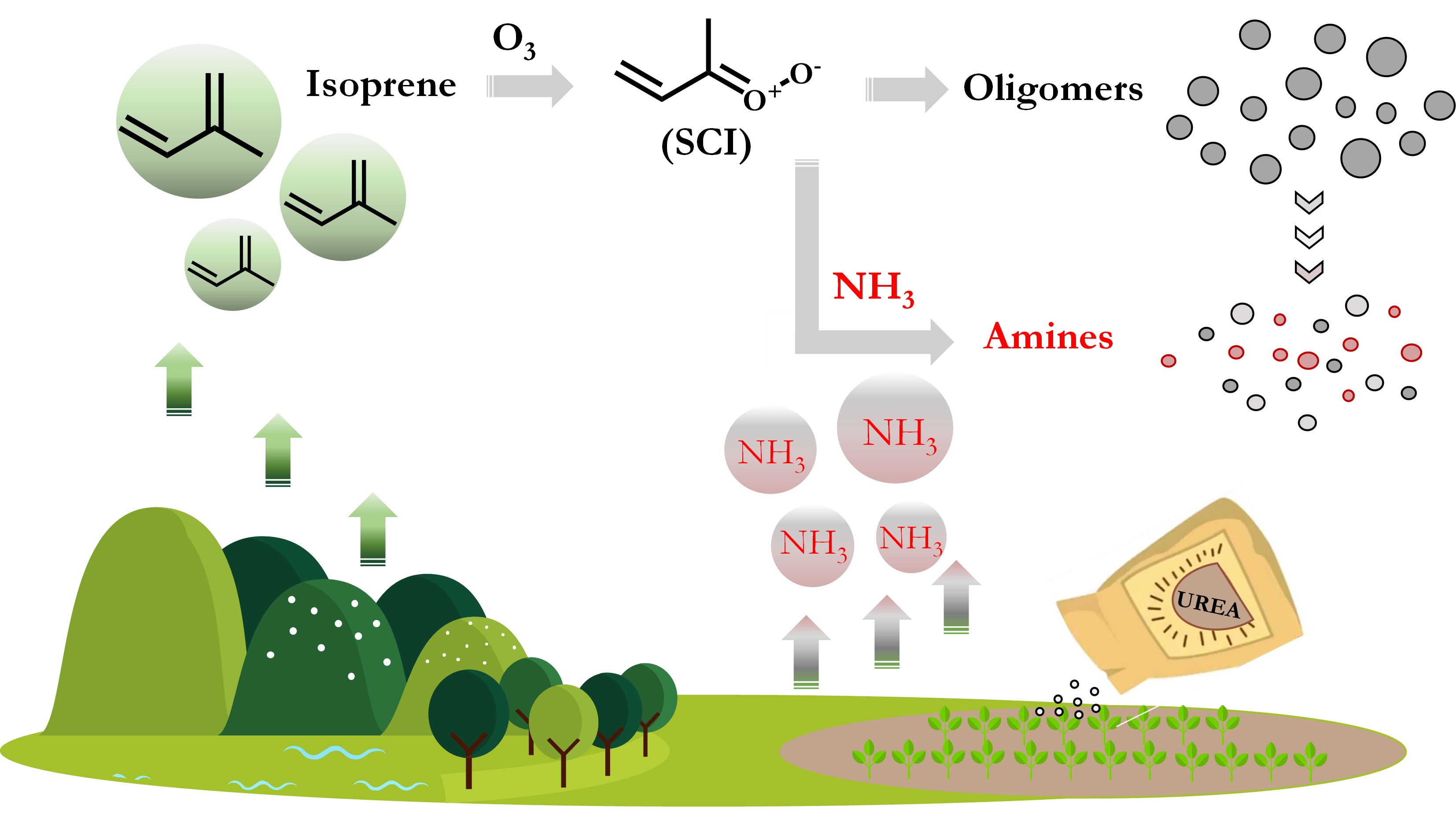
异戊二烯是全球排放量最大的非甲烷挥发性有机物,在大气中具有强反应活性,是全球尺度二次有机气溶胶(SOA)最主要的前体物。氨气(NH3)是大气中含量最丰富的碱性气体,也是大气细颗粒物(PM2.5)中无机盐的主要前体物质。中国是氨气排放的热点区域之一,我国正计划未来削减氨排放,以降低大气PM2.5污染。但是,目前对于异戊二烯和氨在大气转化中的相互作用缺少基本了解。
课题组基于自主研发的气体-气溶胶原位电离技术(GAIS)和轨道阱质谱(Orbitrap-MS),研究了NH3在异戊二烯臭氧化形成SOA中的化学机理。发现NH3可以与异戊二烯臭氧化产物-稳态克氏中间体自由基(SCIs)反应生成一种全新的有机胺分子(C4H9O2N),NH3-SCIs反应通道会显著抑制SCIs的低聚反应,从而极大降低异戊二烯的SOA产率。同时,新生成的有机胺分子还会进一步与有机酸反应生成更多含氮有机物。该研究的重要意义在于:(1)提供了NH3与SCIs反应生成有机胺的直接证据,发现了大气中还原态有机氮生成的新通道;(2)发现氨气可以抑制异戊二烯的SOA产率。这意味着氨气在生物源SOA中的作用比当前的认识更为复杂,有必要对氨减排的潜在环境效应进行深入评估。

论文第一作者为李晓颖博士生,通讯作者贾龙研究员,合作者包括徐永福研究员和潘月鹏研究员。
Li, X.Y., Jia, L.*, Xu, Y.F., Pan, Y.P., 2024. A novel reaction between ammonia and Criegee intermediates can form amines and suppress oligomers from isoprene, Sci. Total Environ., 956, 177389, https://doi.org/10.1016/j.scitotenv.2024.177389
组内消息
2024-09-29
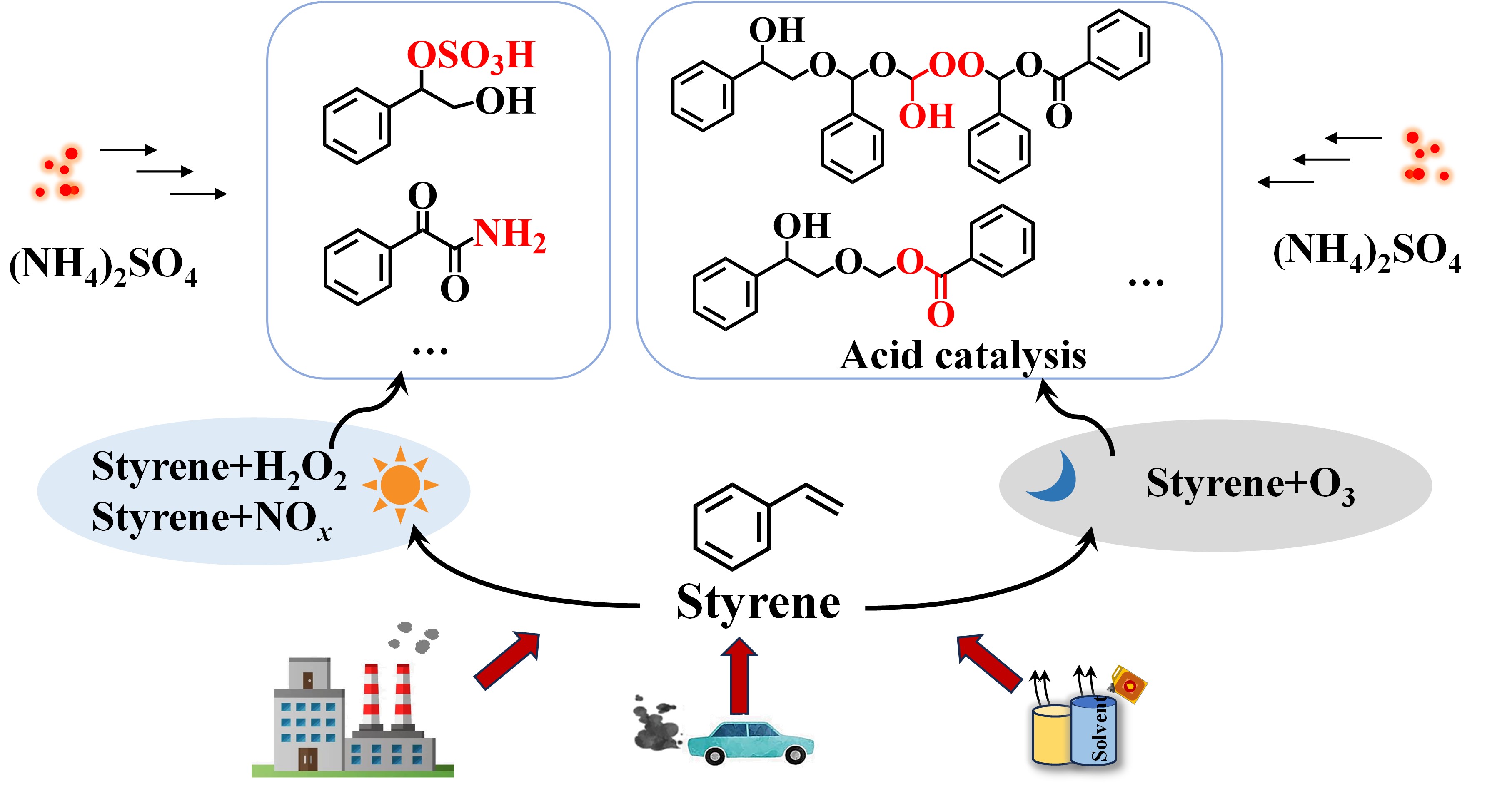
以硫酸铵等无机盐为代表的二次无机气溶胶(SIA)和二次有机气溶胶(SOA)是大气细颗粒物PM2.5的主要组成。由于SOA的成分极其复杂,因而过去的研究往往集中于SOA的生成和演化,很少关注SIA与SOA之间的相互作用。硫酸铵与SOA相互作用可能会改变气溶胶的毒性和光学特性,因此,忽略SIA与SOA之间的交叉反应,会限制我们对PM2.5理化特性的全面了解。
课题组利用实验室模拟和高分辨轨道阱质谱技术,发现烯烃降解过程中克氏中间体自由基之间存在普遍的交叉反应过程,并最新发现硫酸铵与SOA之间也存在着显著的交叉反应。研究团队分析了在不同环境条件下,硫酸铵与苯乙烯氧化产生的SOA之间交叉反应的分子组成,结果表明,铵盐主要通过颗粒相反应生成含氮有机物,而硫酸盐则主要参与形成有机硫酸酯。此外,硫酸铵的存在显著改变了苯乙烯克氏中间体生成低聚物的反应路径。这项研究不仅加深了我们对SOA形成机制的认识,突显了SIA与SOA分子间相互作用的重要性。硫酸铵与SOA相互作用会生成含氮和含硫有机物这一发现,对于揭示城市大气中PM2.5的毒性和光学特性具有重要的科学意义。
上述研究成果近期发表于期刊《Science of the Total Environment》,论文第一作者为于姗杉博士,通讯作者贾龙研究员,合作者包括徐永福研究员和潘月鹏研究员。
Yu, S.S., Jia, L.*, Xu, Y.F., Pan, Y.P., 2024. Molecular interaction between ammonium sulfate and secondary organic aerosol from styrene. Sci. Total Environ., 954,176414, https://doi.org/10.1016/j.scitotenv.2024.176414
组内消息
2024-04-08
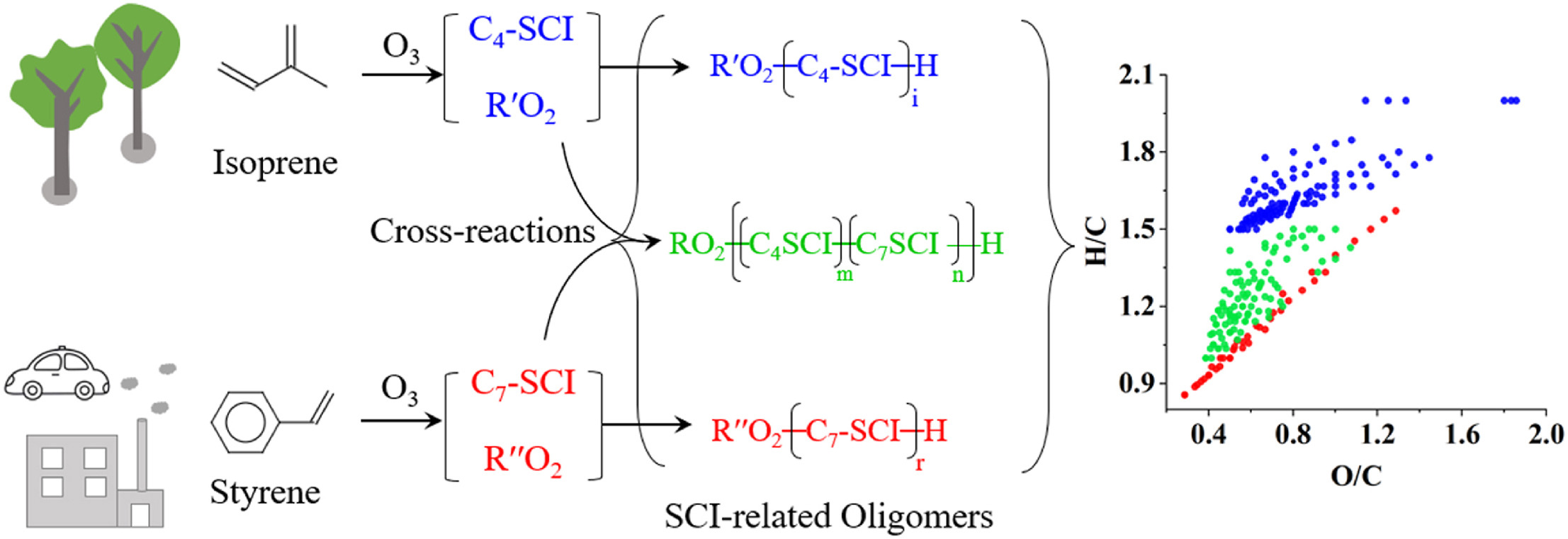
二次有机气溶胶(SOA)是大气细颗粒物的重要组成部分,会对环境、气候和人体健康产生影响。苯乙烯(styrene)和异戊二烯(isoprene)是人为和自然源SOA的重要前体物,在大气环境中广泛存在,但它们间的交叉反应并未受到关注。课题组基于烟雾箱模拟实验和高分辨轨道阱质谱技术(Orbitrap MS),研究了由苯乙烯和异戊二烯产生的克氏自由基中间体(Criegee SCIs)之间的交叉反应,以及交叉反应对SOA的形成和物理化学性质的影响规律。发现来自苯乙烯的特征C7-SCI和异戊二烯的特征C4-SCI可以发生交叉反应,并导致混合体系的SOA产率低于单一苯乙烯-O3体系,但高于单一异戊二烯-O3体系。同时在外场环境采集的细颗粒物中也发现了SCI相关的交叉产物。该研究证实SCI在决定苯乙烯-异戊二烯混合体系SOA的生成和理化性质方面的起着关键作用,说明在城市环境中,人为和自然源挥发性有机化合物(VOCs)的相互作用会显著改变SOA的理化性质,突显了交叉反应的重要性。
Yu, S.S., Jia, L.*, Xu, Y.F., Pan, Y.P., 2024. Oligomer formation from cross-reaction of Criegee intermediates in the styrene-isoprene-O3 mixed system. Chemosphere, 349, 140811, https://doi.org/10.1016/j.chemosphere.2023.140811
- « PREV
- 1
- 2
- 3
- 4
- 5
- 6
- ...
- 19
- NEXT »