New paper about effects of inorganic salts on SOA formation from acetone is published in AE
组内消息 2017-09-28
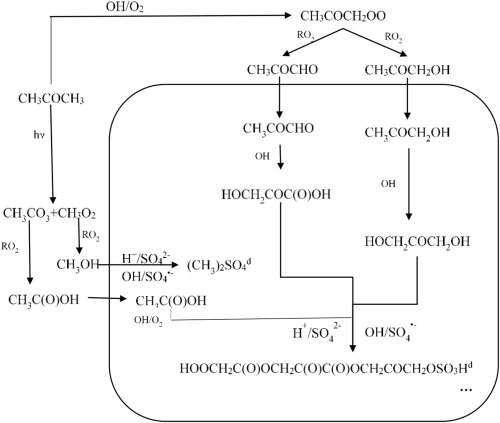
Effects of inorganic seeds on secondary organic aerosol formation from photochemical oxidation of acetone in a chamber
Highlights
•Acetone is confirmed to be one of the sources of SOA.
•Participation of inorganic seeds into acetone SOA is determined.
•Chemical compositions and formation mechanism of acetone SOA are proposed.